Macau’s health chief says it will begin administering Sinopharm jabs on Tuesday, despite a lack of Phase 3 clinical data, while Hong Kong officials and advisers insist the city will not cut corners in its own approval process.
A leading government health adviser has said Hong Kong will not cut corners when it comes to approving Covid-19 vaccines developed on the mainland, even as neighbouring Macau received its first batch of 100,000 doses from Beijing on Saturday.
In a ceremony welcoming the arrival of the mainland-produced Sinopharm jabs, Macau’s health chief announced that the city would commence vaccination for priority groups, including medical staff, starting on Tuesday. In light of the lack of comprehensive Phase 3 data on the vaccine, he also vowed to get the first jab himself as a display of his faith in the drug.
“I am confident in the vaccines,” said Dr Lei Chin-ion, director of Macau’s Health Bureau. “And soon after the medics get vaccinated around the Lunar New Year, we will open for public registration.”
 Workers in Macau unload the first batch of Sinopharm vaccines from Beijing on Saturday.
Workers in Macau unload the first batch of Sinopharm vaccines from Beijing on Saturday.
Hong Kong, meanwhile, is still waiting for Sinovac – another mainland-based vaccine manufacturer from which it has secured 7.5 million doses – to provide more clinical information so its product can be approved. Local health authorities have also insisted it provide data published in an international journal for assessment, but Sinovac has said it would take more time to compile all the necessary information for peer review.
In light of the delay, the government said in a statement on Friday night that Sinovac would provide its raw Phase 3 data from Brazil – the same set it recently submitted to the WHO – so the expert panel could begin its assessment while waiting for the peer-reviewed research. Peer review, the statement noted, would indicate a higher level of objectivity in the results, and acceptance by the broader scientific community.
Government advisers in Hong Kong defended the “necessary move” of holding out for the full, vetted data, noting that the city and Macau had different standards for approving vaccines, and that the local government and experts would not sacrifice safety for the sake of speed.
Dr David Hui Shu-cheong, who advises the government on Covid-19 vaccines, told the Post on Saturday that Hong Kong followed international standards when approving vaccines, and maintained that providing Phase 3 data was a must.
“We have to see the [effective] age range of the vaccines, whether they have caused comorbid illnesses in patients, how high are the levels of antibodies and whether they can help to prevent the variant virus,” he said.
Hui noted that, so far, neither Sinovac nor Sinopharm had provided such data to Hong Kong or Macau. And based on the high standard of information requested by Hong Kong officials, he added, it was understandable that the process here would take longer.
Hui maintained the government and the board were trying to get the data as fast as they could, but promised they would not sacrifice standards.
“We believe Sinovac will still publish the data in medical journals, but that might take longer than expected, thus we could still start analysing their data while waiting for the publication,” he said.
Hong Kong Chief Executive Carrie Lam Cheng Yuet-ngor has also defended the city’s approach as compared to Macau’s. At a press conference on Tuesday, she said the central government understood Hong Kong had its own legal framework for emergency use of vaccines, including on the evaluation of data by experts before a drug could be recommended to the secretary for food and health.
Hong Kong has already struck deals to purchase 22.5 million doses of vaccines, with 7.5 million coming from each of three suppliers: Sinovac Biotech, AstraZeneca, and Pfizer, which co-developed its vaccine with the German company BioNTech.
The Sinovac jabs were originally intended to be the first to arrive for the government’s planned universal vaccination programme.
Authorities have already approved the Pfizer-BioNTech vaccine, and the first shipments are due to arrive by late February.
















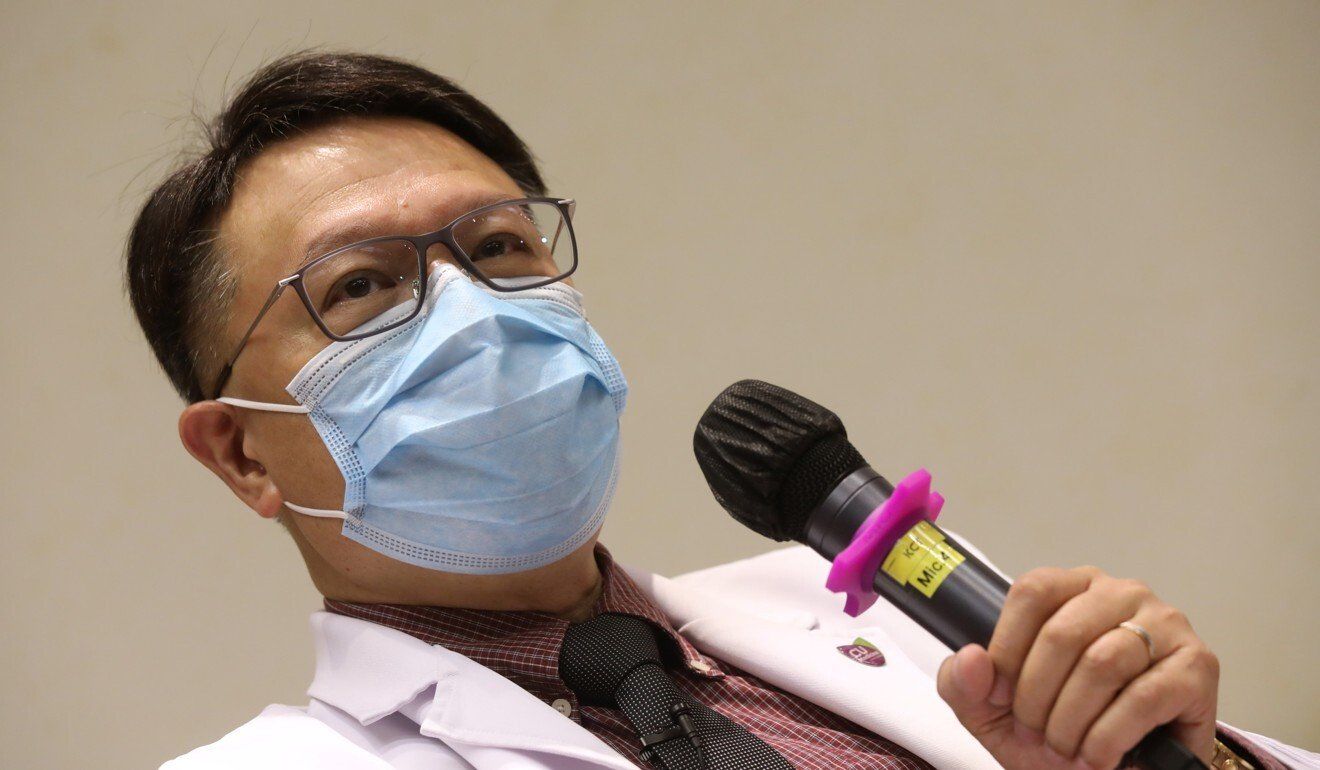 Dr David Hui Shu-cheong, an adviser to the Hong Kong government on
Dr David Hui Shu-cheong, an adviser to the Hong Kong government on